 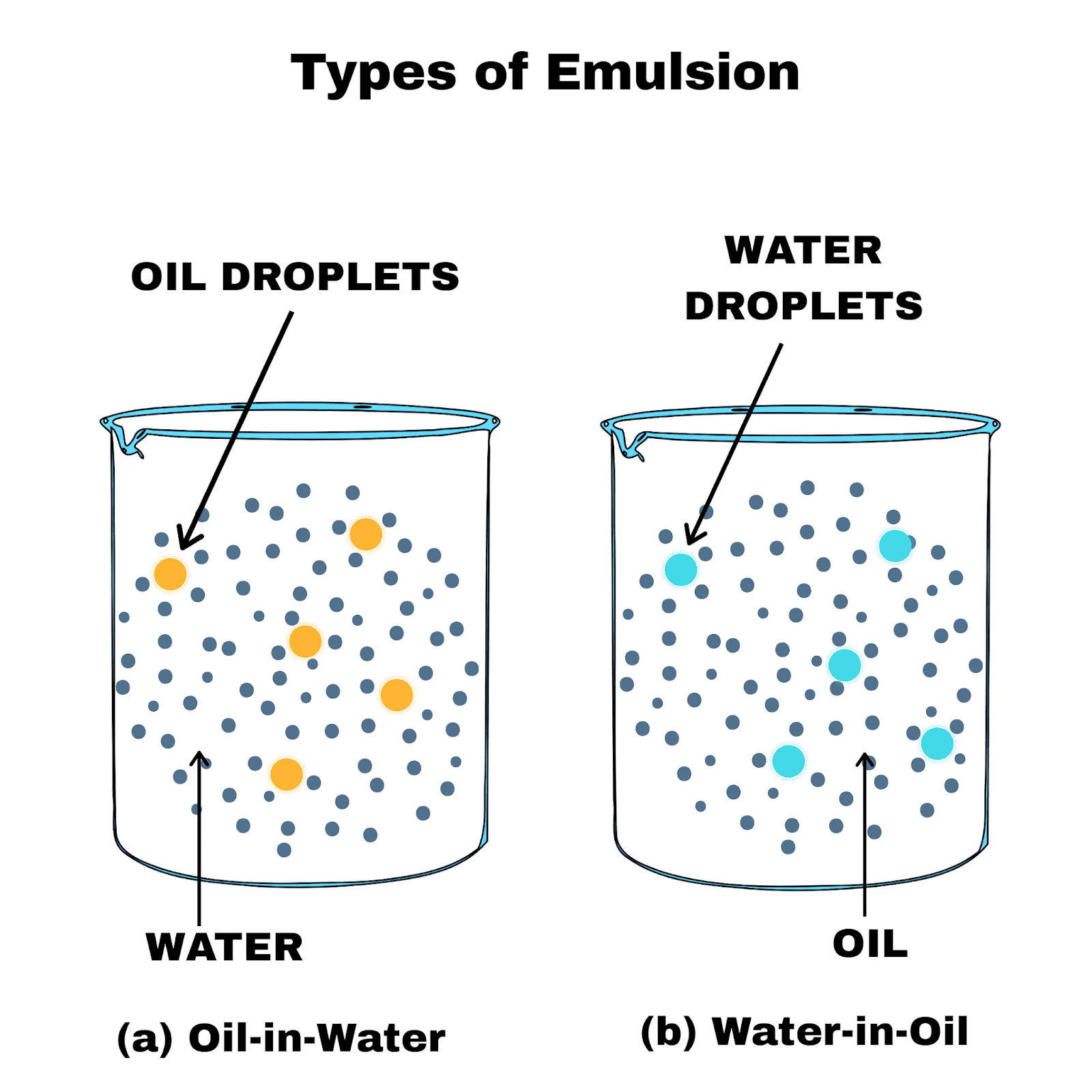
The particle size within emulsions is critically important in many applications one in particular is intravenous (IV) infusions. The zeta potential of an emulsion indicates the likelihood of the droplets to aggregate and is thus related to the overall stability of the emulsion. The droplet size of an emulsion is important to ensure the ideal look and feel of an emulsion as well as the required functionality such as the rate of drug delivery of an intravenous (IV) emulsion (Figure 1) or the dose uniformity of an inhaled drug. Two of the most commonly used are dynamic light scattering (DLS), which measures the particle size and size distribution of the emulsified droplets, and electrophoretic light scattering (ELS) which measures the droplet charge/ zeta potential. There are a number of scientific techniques useful for characterizing emulsions. Thus, it is critically important to be able to accurately and quickly measure these properties of emulsions. Specifically, the size and charge of the emulsified droplets directly affect stability, taste, safety, look and feel, and function. Examples of common emulsifiers include mustard, egg yolk, soaps, and other surfactants.Įmulsions are often perceived as something simple, macroscale that can be seen, felt, and even tasted but in reality it is the nanoscale properties of the droplets dispersed in an emulsion that are the main contributors to the emulsion’s bulk properties. Emulsifiers are structurally and functionally similar to surfactants in that they have a long hydrophobic chain that extends into the oil layer and a polar head-group that interacts with the aqueous layer. That being said, stable emulsions can be generated by adding an emulsifier, which is a chemical that helps to keep the two components mixed. However, because there is a natural tendency for oil and water to separate, most emulsions are not stable over time and their components will separate out into two layers.  They can also be formed by sonication, which uses ultrasonic waves to agitate dispersions and cause them to mix, as well as by homogenization, which is commonly used to process milk so that fat droplets remain dispersed and do not float to the top. Similar to a vinaigrette salad dressing, emulsions can be formed, at least temporarily, by rapidly shaking or stirring an oil and water mixture together. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
